Evidence of Efficacy
In Vivo Survival Study
ACT-903
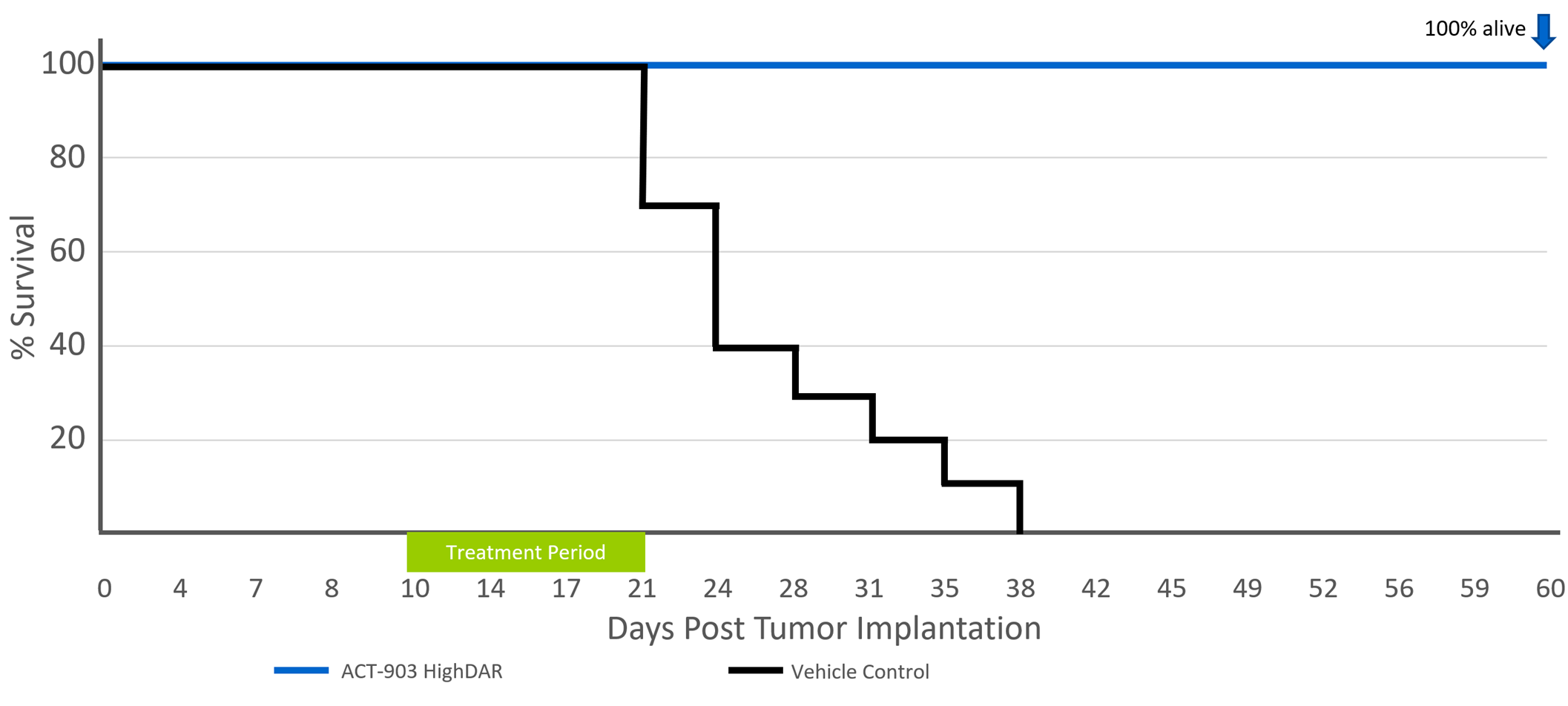
Effect of ACT-903 Conjugate on Survival
COLO-205 Xenograft Model (10 per Group)
Kaplan-Meier Survival – following 10 days of treatment – all untreated animals are dead at Day 30 (50% dead at day 24) while 100% of treated animals are alive at Day 60.
Highlights From The Study
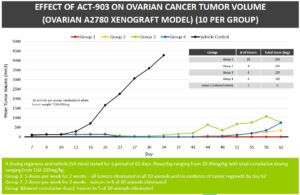
- Statistically significant reduction in tumor volume was observed in all treatment groups compared to control beginning at Day 17. In one of the conjugate groups, tumor reduction continued following treatment discontinuation with tumor volumes falling below the limit of detection in 9 of 10 animals
- 100% survival in ACT-903 group at day 60, compared to 0% survival in the control group by day 38
- After a single IV dose, biodistribution study of conjugate showed excellent tumor targeting with maytansine and metabolite accumulation and undetectable bone marrow toxicity
- No signs of toxicity were observed in treated mice
ACT has identified the ACT-903 conjugate with the strongest efficacy profile and will advance the program into further studies to support a Phase 1 clinical trial.
ACT-901
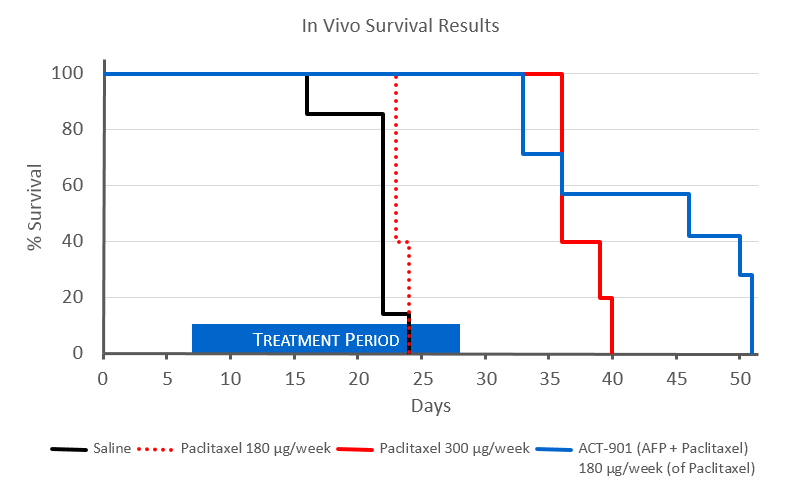
In this study several groups of mice were treated with various doses of paclitaxel (Taxol®) or ACT-901 (AFP + paclitaxel). The black line shows the control group that was given only saline. All mice were dead by day 24.
The 2 red lines show survival of mice given low (dotted red line) or high (solid red line) of paclitaxel. Low dose paclitaxel had no
effect and did not improve survival rates.
The same low paclitaxel dose (blue line) delivered as ACT-901 (AFP + paclitaxel) significantly improved survival and was even better than a high dose paclitaxel.
There were 7 mice per group in this study. Low dose paclitaxel and ACT-901 (AFP + paclitaxel) were given every other day for 3 weeks at
the same dose of paclitaxel, while in high dose paclitaxel group treatment was daily for 3 weeks.
Mice in the high paclitaxel group (solid red line) showed signs of severe toxicity, with weight loss and lack of normal grooming
behaviour and excessive irritability. The ACT-901 (AFP + paclitaxel) group exhibited no signs of toxicity.
There was also significant vein sclerorsing in the high paclitaxel group but not in the ACT-901 (AFP + paclitaxel) group.
UHN, Princess Margaret Cancer Centre
In Vivo Xenograft Study
ACT-903 (AFP + Linker + Maytansine)
Presented at the European Society of Clinical Oncology (September 2021)
Title: AFP-Maytansine Conjugate – a Novel Targeted Cancer Immunotherapy
Abstract Number: 523P
The poster titled, “AFP-Maytansine Conjugate – a Novel Targeted Cancer Immunotherapy,” highlights preclinical data from an animal study conducted with Southern Research Institute (SRI) evaluating ACT-903, a novel protein drug conjugate using a proprietary version of recombinant human AFP, combined with a proprietary chemical linker and proprietary maytansine toxin.
Effect of ACT-903 Conjugate on Tumor Volume
COLO-205 Xenograft Model (10 per Group)
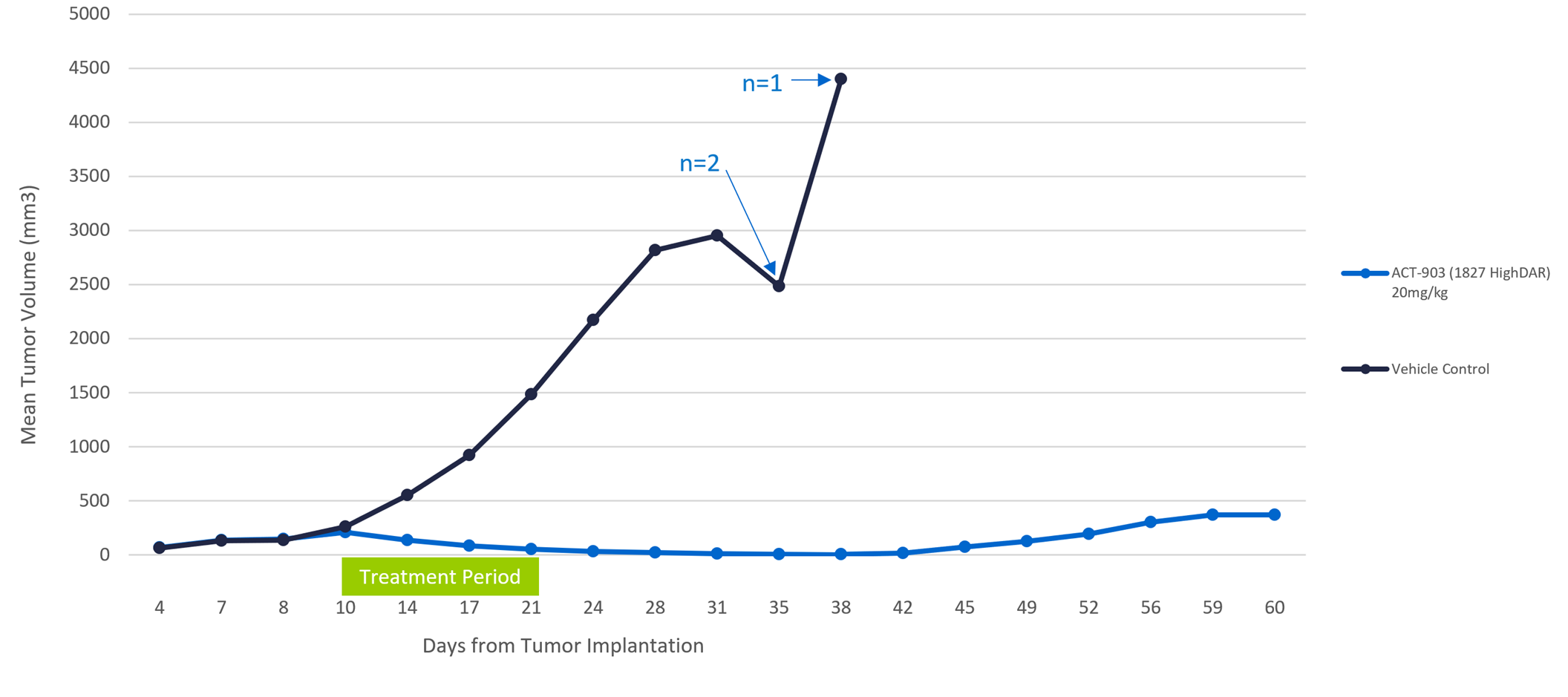
During the study, four novel AFP-maytansine conjugates of differing drug-protein ratios and slightly different linker structures were administered intravenously (IV) to mice bearing human colon carcinoma (COLO-205) xenografts, at doses previously determined to be safe. Seven days after implantation, mice with tumors greater than 150 mm3 were randomized to receive control or one of the four conjugates (10 animals/ group). Best response group shown below. Animals were treated daily for two weeks with two days of rest after five doses and tumor volume was assessed twice weekly for 60 days following implantation.
ACT-901
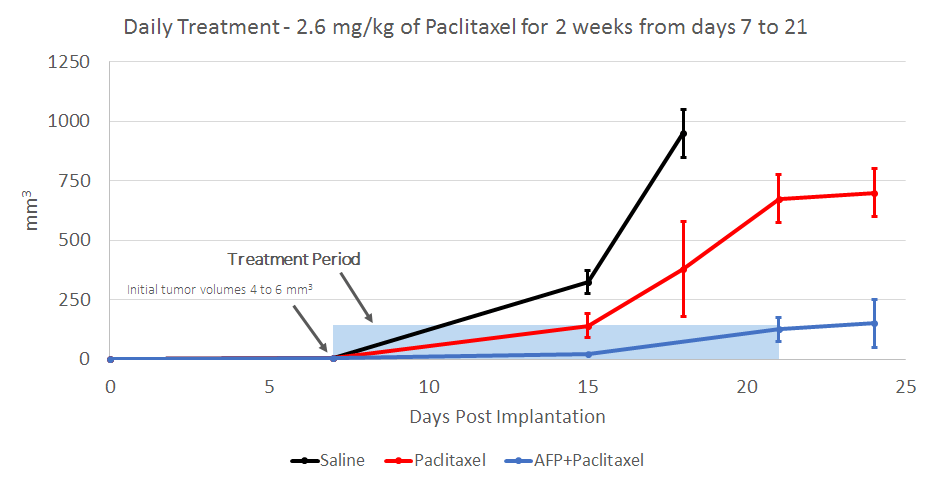
In this study 3 groups of mice were treated with either saline or with equal doses of paclitaxel (Taxol®) delivered either as a conventional
paclitaxel formulation (red line) or carried by AFP (ACT-901) (blue line). The graph illustrates that tumor growth is significantly reduced when paclitaxel is delivered by AFP (ACT-901) compared to the
same dose of free paclitaxel.
UHN, Princess Margaret Cancer Centre
In Vivo MDSCs Study | PDX Model
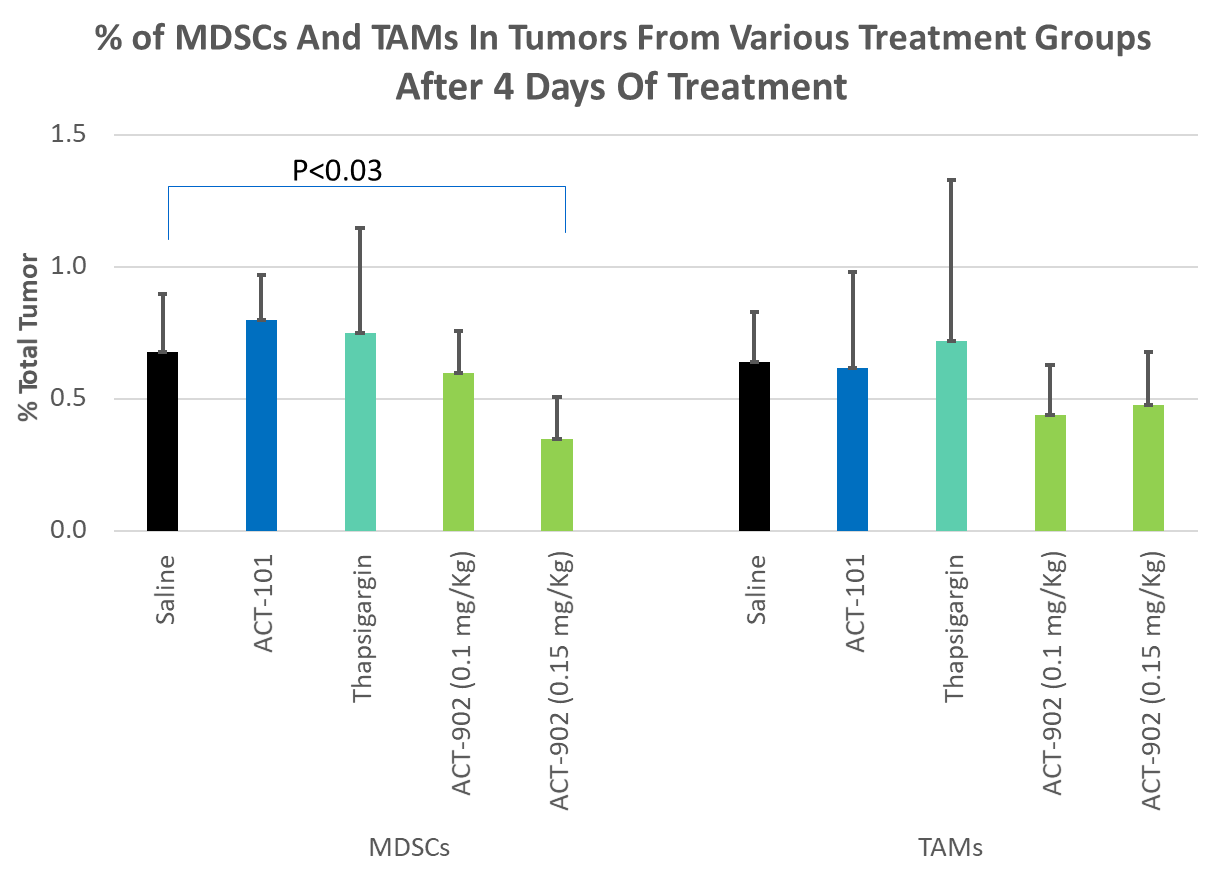
ACT-902 causes significant reduction in Myeloid Derived Suppressor Cells (MDSCs) that play a major role in protecting cancer cells from host immune system. Thus AFP conjugates are likely to have both, direct cytotoxic effect on cancer cells and immune check point inhibition effect without toxicity.
UHN, Princess Margaret Cancer Centre
In Vitro Study
Overcoming Multidrug Resistance In Human Ovarian Cell Line
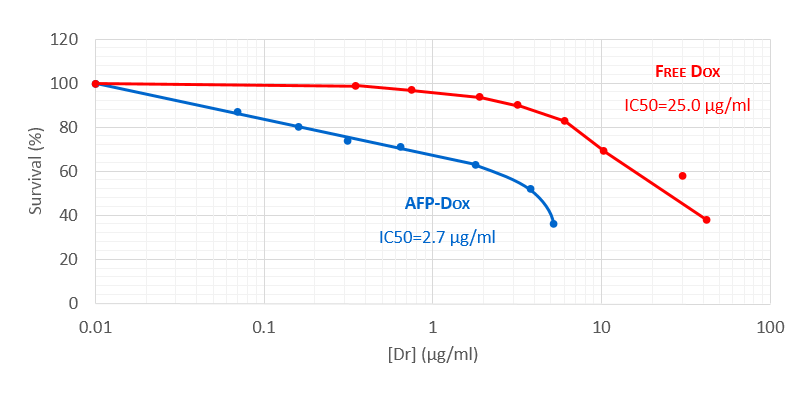
A study on chemo-resistant ovarian carcinoma cell line SKVLB demonstrated that is takes 10 times more dox to kill 50% of the cells in the resistant cell line than with AFP-Dox which seems to be able to overcome the resistance by binding to receptors found on the resistant cells and transporting dox directly into the cell bypassing the cell membrance pumps.
Moskaleva et al. (1997), Cell Biol. Internat., 21, 793-79